Abstract
Background: Chemotherapy combined with anti-CD20 antibodies is the current standard treatment for patients with symptomatic advanced stage follicular lymphoma (FL). We have previously shown that the EZH2 gene mutation status was predictive of differential efficacy of the chemotherapy backbone within the GALLIUM trial. Specifically, EZH2 mutations, found in 22% of cases (93/418), were associated with longer progression-free survival (PFS) in patients who received CHOP/CVP regimens (with either Rituximab (R) or Obinutuzumab (G)), but were not predictive in patients who received Bendamustine-based regimens (Jurinovic, ASH 2019). In an earlier study, we defined a gene expression profile (GEP) signature which was significantly associated with EZH2 gene mutation status (Pastore, Lancet Oncol 2015). Here, we wanted to explore the predictive utility of this EZH2 GEP signature within the GALLIUM trial.
Methods: GALLIUM (NCT01332968) enrolled 1202 patients with FL randomized to either receive R- or G-based therapy (Marcus, NEJM 2017). The chemotherapy consisted either of CHOP, CVP or Bendamustine according to a center-level decision. Inclusion criteria comprised previously untreated FL, advanced stage or stage II with bulky disease, and ECOG performance status 0-2. All patients required treatment according to the GELF criteria. Here, we analyzed all 134 cases with available DNA and transcriptome sequencing data.
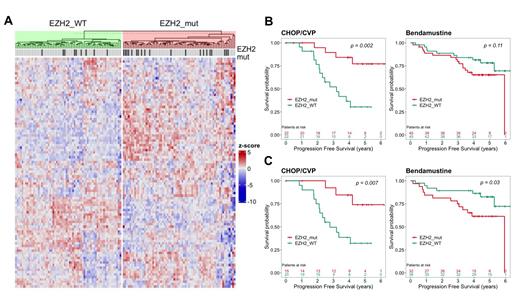
Results: Of the 129 genes from the original EZH2 GEP signature, 96 (74%) were evaluable in our transcriptome data. We used the unsupervised k-means algorithm to identify 2 GEP clusters, EZH2_mut and EZH2_WT, comprising 67 patients each (Fig A). Patients in the EZH2_mut cluster had significantly longer PFS with CHOP/CVP-based regimens (HR = 4.99, P = 0.002), while the EZH2 GEP signature was not predictive in patients receiving Bendamustine-based treatment (Fig B). As expected, EZH2 mutations were significantly enriched in the EZH2_mut cluster (30% vs 13%; P = 0.034). Of note, 47 cases in the EZH2_mut cluster had no detectable EZH2 mutation. We wanted to test, whether the predictive utility of the EZH2 GEP signature was maintained in cases without EZH2 mutations. Indeed, patients without EZH2 mutations grouped into the EZH2_mut cluster also had longer PFS with CHOP/CVP-based regimens (HR = 4.74, P = 0.007), whereas the ones grouped into the EZH2_WT cluster had longer PFS with Bendamustine-based regimens (HR = 0.36, P = 0. 03) (Fig. C). To explore the genetic determinants of the EZH2 GEP signature in cases without EZH2 mutations, we analyzed the available DNA sequencing data, considering recurrent non-silent gene mutations and copy-number alterations (CNA) with frequencies of 10% in either cluster. As expected, gain of chromosome 7 (chr7), which contains the EZH2 gene, was enriched in the EZH2_mut cluster (21% vs 7%, P = 0.04). Cases with chr7 gain had significantly increased EZH2 mRNA expression levels as compared to cases without EZH2 CNA or mutations (P < 0.0001). Furthermore, univariate analysis revealed enrichment of mutations in TNFRSF14 (49% vs 24%), BCL7A (28% vs 8%), GNA13 (15 vs 7%), SOCS1(19 vs 8%), and HVCN1 (13 vs 7%) in the EZH2_mut cluster, but none of these associations were significant after correction for multiple testing. Finally, we wanted to better delineate the transcriptional phenotype of the EZH2 GEP signature in cases that lack EZH2 mutations. GSEA analysis revealed that the EZH2_mut cluster was strongly enriched for the hallmark signatures mTORC1 signaling, MYC and E2F target genes. Furthermore, we used CIBERSORT to estimate the immune cell composition of the tumor microenvironment (TME). The TME compositions of FL in the EZH2_mut cluster lacking EZH2 mutations more closely resembled FL with EZH2 mutations, most notably for higher T follicular helper (TFH) cell (P = 0.003) and macrophage M0 scores (P < 0.001) as compared to FL in the EZH2_WT cluster lacking EZH2 mutations.
In conclusion we show that an EZH2 GEP signature is (i) significantly associated with EZH2 gene mutation status, (ii) also observed in a subset (47/105, 45%) of cases that lack EZH2 mutations but harbor other genetic alterations (including chr7 gain/EZH2 overexpression), (iii) associated with a distinct TME composition enriched for TFH cells and M0 macrophages, and (iv) predictive of differential efficacy of chemotherapy-backbones in combination with Rituximab or Obinutuzumab.
Bolen: F. Hoffmann-La Roche: Current equity holder in publicly-traded company; Genentech: Current Employment. Knapp: F. Hoffmann-La Roche Ltd: Current Employment. Bottos: F. Hoffmann-La Roche Ltd: Current Employment, Current equity holder in publicly-traded company. Richter: HTG Molecular Diagnostics, Inc.: Current Employment, Research Funding. Fitzgibbon: Epizyme: Research Funding. Klapper: Takeda: Consultancy, Research Funding; Amgen: Research Funding; Regeneron: Consultancy, Research Funding; Roche: Consultancy, Research Funding. Davies: Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel to scientific conferences, Research Funding; Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel to scientific conferences, Research Funding; Kite: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Honoraria, Research Funding; Karyopharm Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Acerta Pharma/AstraZeneca: Honoraria, Research Funding; ADC Therapeutics: Honoraria, Research Funding; BioInvent: Research Funding; Incyte: Membership on an entity's Board of Directors or advisory committees. von Bergwelt: MSD Sharpe & Dohme: Honoraria, Research Funding, Speakers Bureau; Mologen: Honoraria, Research Funding, Speakers Bureau; Kite/Gilead: Honoraria, Research Funding, Speakers Bureau; Astellas: Honoraria, Research Funding, Speakers Bureau; BMS: Honoraria, Research Funding, Speakers Bureau; Roche: Honoraria, Research Funding, Speakers Bureau; Novartis: Honoraria, Research Funding, Speakers Bureau; Miltenyi: Honoraria, Research Funding, Speakers Bureau. Nielsen: F. Hoffmann-La Roche Ltd: Current Employment, Current equity holder in publicly-traded company. Dreyling: Bayer HealthCare Pharmaceuticals: Consultancy, Research Funding, Speakers Bureau; Celgene: Consultancy, Research Funding, Speakers Bureau; Astra Zeneca: Consultancy, Speakers Bureau; BeiGene: Consultancy; Novartis: Consultancy, Speakers Bureau; Genmab: Consultancy; Amgen: Speakers Bureau; Gilead/Kite: Consultancy, Research Funding, Speakers Bureau; Incyte: Consultancy, Speakers Bureau; Janssen: Consultancy, Research Funding, Speakers Bureau; Roche: Consultancy, Research Funding, Speakers Bureau; Abbvie: Research Funding. Hiddemann: Janssen: Research Funding; F. Hoffmann-La Roche: Membership on an entity's Board of Directors or advisory committees, Research Funding. Unterhalt: Roche: Research Funding. Weigert: Janssen: Speakers Bureau; Epizyme: Membership on an entity's Board of Directors or advisory committees; Roche: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal